- Developing NRX-101, an FDA-Designated Investigational Breakthrough Therapy for Suicidal Treatment-Resistant Bipolar Depression and Chronic Pain.
- Designed to Help Address the Needs of Over 13 Million Americans who Seriously Consider Suicide Each Year (CDC).
- D. Boral Analyst Report on NRXP $34 Price Target.
- Type C Meeting with the FDA Demonstrates a Path to New Drug Application with Real World Data and Broader Proposed Indication for NRX-100 (ketamine).
- NRXP Applauds Presidential Initiative to Accelerate Approval of Psychedelic Medications to Treat Depression, PTSD, and Suicidality.
- Positive FDA Review Letter Addressing Drug Quality Requesting Only “Minor” Administrative Changes.
- In process of Submitting a New Drug Application to Align the Labeling of Ketamine with its Use in Treating Depression and Suicidality.
- President Trump Signed Executive Order to Accelerate Treatments for Mental illness on April 18, 2026 with Support for Psychedelic Medications.
- Robotic-Enabled Transcranial Magnetic Stimulation Combined with Neuroplastic Therapy for Military and First Responder Applications
- Dennis K. McBride, PhD Appointed President of New Defense Systems Subsidiary.
- Combined Neuroplastic Therapy including NRX-101 and Robotic Transcranial Magnetic Stimulation in Military and First-Responder Setting.
- FDA Labeling Alignment for NRx’s Preservative-Free Ketamine Application.
- Strategic Partnership with Emobot to Integrate AI-Powered “Depression Thermometer” Across Interventional Psychiatry Network.
- 2025 Results Included Reduction in Operating Expenses with $7.8 Million Cash on Hand. Adequate Resources to Support Operations Through 2026.
- FDA Bioequivalence Determination by Office of Generic Drugs for NRx’s Preservative-Free Ketamine Application.
- Anticipated FDA Approval of at Least One NRXP New Drug – Preservative Free Ketamine Under an ANDA—During Q3 2026.
- Published Results Have Shown 87% Clinical Response to Non-Invasive Transcranial Magnetic Stimulation with Neuroplastic Medications.
- Clinical Leadership by Physicians Trained at Harvard, Johns Hopkins, Georgetown, and Other Leading Universities.
- On Path to Application for New Drug Approval of NRX-100 Under Fast Track Designation Based on Existing Clinical Trial Data and Real World Evidence.
- NRXP Will Seek Broader Indication to Serve Patients with Treatment-Resistant Depression Who May Have Suicidality Rather Than the Subset with Suicidality.
- 70,000 Patient Data on Real World Use of Ketamine for Treatment of Suicidal Depression to be Submitted to the FDA in Support of NRX-100 Approval
- NRXP and neurocare Group AG Announce Joint Offering of Neuroplastic Therapy Targeting Depression, PTSD and Other Mental Afflictions.
- Elimination of All Balance Sheet Debt.
- Current Worldwide Generic Ketamine Market Estimated at $750 Million Per Year.
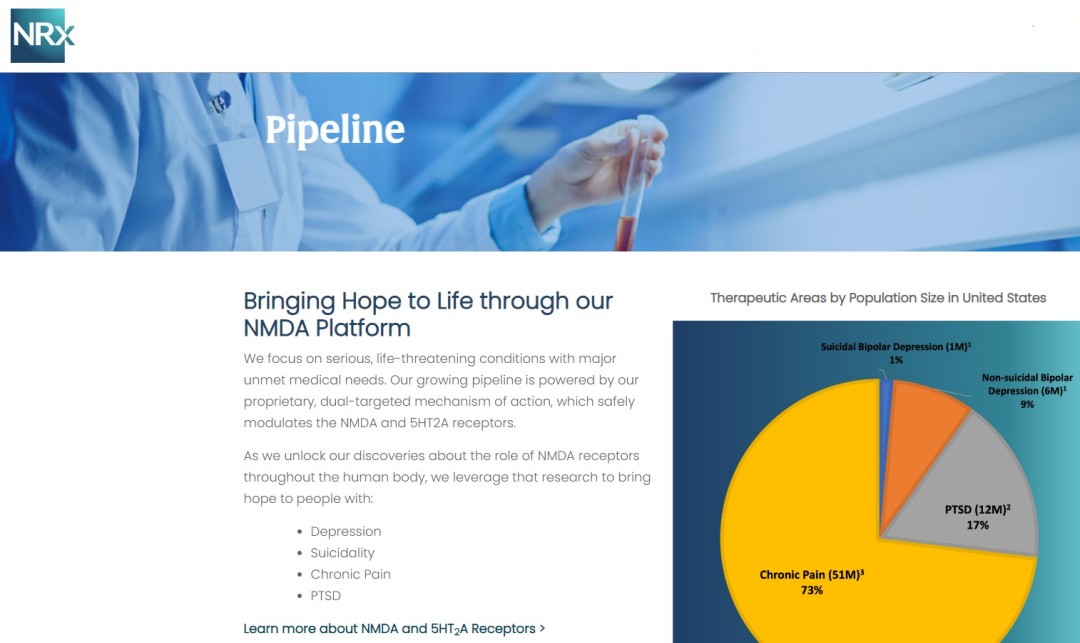
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. NRXP is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain.
NRXP has partnered with Alvogen Pharmaceuticals around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRXP is working on a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRXP was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
D. Boral has issued an Analyst Report on NRXP with a Buy and $34 Price Target. The full report may be accessed at this direct link: https://www.nrxpharma.com/wp-content/uploads/2025/11/HOPE-Therapeutics-NRXP-Executes-Florida-Roll-out-of-Ampa-O.pdf
Positive FDA Office of Generic Drugs Feedback on Preservative-Free Ketamine Program
On April 22nd NRXP announced the receipt of a positive Discipline Review Letter from the FDA Office of Generic Drugs and the completion of a supportive meeting with Generic Drug Leadership. The Discipline Review Letter entitled “Quality” covers the areas of Drug Substance, Drug Product, Manufacturing, and Microbiology. The letter requests only administrative changes and updates to prior stability data, all of which are identified as “Minor.” This positive review letter follows the previously announced favorable bioequivalence determination from the FDA Office of Generic Drugs on March 17, 2026, and represents a separate review discipline within the ANDA process.
NRXP additionally conducted a meeting with leadership of the FDA Office of Generic Drugs in which the national priority around expediting approval of ketamine, now documented in the President’s Executive Order, was recognized by FDA leadership. The Executive Order -- ACCELERATING MEDICAL TREATMENTS FOR SERIOUS MENTAL ILLNESS calls for expedited approval of drugs to treat severe depression and suicidality. The approval of the preservative-free ketamine ANDA is particularly important because of supply shortages documented by physicians who seek to obtain ketamine for use in the clinic setting. FDA leadership expressed support for addressing the remaining aspects of the current ANDA application within the current review cycle that targets approval in Summer 2026.
NRXP presentation of ketamine differs from existing products in that it does not contain a known toxic preservative, Benzethonium Chloride. This preservative is no longer allowed to be included in new drugs and according to FDA policy cannot even be included in hand cleansers and topical antiseptics.
Presidential Initiative to Accelerate Approval of Psychedelic Medications to Treat Depression, PTSD, and Suicidality
On April 20th NRXP welcomed the Executive Order: ACCELERATING MEDICAL TREATMENTS FOR SERIOUS MENTAL ILLNESS, signed by President Trump on April 18, 2026. In the Order, the President notes that, “It is the policy of my Administration to accelerate innovative research models and appropriate drug approvals to increase access to psychedelic drugs that could save lives and reverse the crisis of serious mental illness in America.”
The order directs the Commissioner of the US Food and Drug Administration to provide Commissioner’s National Priority Vouchers (CNPV) to appropriate psychedelic drugs that have received a Breakthrough Therapy designation and are in accordance with the criteria of the National Priority Voucher Program. NRXP has applied for a CNPV in support of its upcoming New Drug Application for NRX-100.
The Presidential order further directs the Department of Health and Human Services and FDA to collaborate with the Department of Veterans Affairs and the Private Sector to increase clinical trial participation, data sharing, and real-world evidence generation regarding psychedelic drugs …to facilitate the timely evaluation and approval of drugs that meet standards for approval under the Federal Food, Drug, and Cosmetic Act. This aspect of the executive order is expected to facilitate NRXP proposed use of Real World Evidence and already-completed federally-funded clinical trials in support of drug approval.
Advancing Robotic-Enabled Transcranial Magnetic Stimulation Combined with Neuroplastic Therapy for Military and First Responder Applications
On April 15th NRXP announced the incorporation of NRx Defense Systems, Inc., a Florida-based Research and Development subsidiary. The subsidiary is charged with development of neuroplastic treatments that combine D-cycloserine with Transcranial Magnetic Stimulation (TMS) under robotic-enabled neuro-navigation, using already developed technology.
The NRXP NRx Defense Systems will be led by Dr. Dennis McBride, whose career spans the intersection of CNS disorders, neuroplastic therapy, and robotics. Dr. McBride has held leadership roles in the Office of the Secretary of Defense, including as Director of the SECDEF Acquisition Innovation Research Center. Dr. McBride has transitioned multiple biomedical and human factors technologies to private industry, with independent estimates of annual revenue enablement ranging from $15 billion to nearly $40 billion.
Combined Neuroplastic Therapy for depression and PTSD is an increasing area of focus within military organizations because of the recognized five-fold increased risk in front-line troops and first responders. Accordingly, rapid and efficient treatment of these conditions becomes not only a critical healthcare issue, but also a force-readiness issue, in that those on antidepressants are not combat-deployable. A key objective of the NRXP Defense Systems initiative is to develop robotic and artificial intelligence-enabled solutions that can be forward-deployed within military organizations without relying on a new layer of skilled medical professionals.
The NRXP prototype is being developed in partnership with Zeta Surgical, Inc. NRXP anticipates unveiling the prototype robotic-enabled TMS technology with neuroplastic therapy at the 14th Annual meeting of the Clinical TMS Society (CTMSS) from June 4-6, 2026.
FDA Labeling Alignment for NRx’s Preservative-Free Ketamine Application
On April 6th NRXP announced that it has received a letter from the Labeling Program of the FDA Office of Generic Drugs with only minor formatting changes to the proposed label for the Company's preservative-free ketamine product. The determination is deemed preliminary until final supervisory review of the NRXP Abbreviated New Drug Application with anticipated approval in Summer 2026.
This determination by FDA is meaningful in that the NRXP product has the potential to substantially augment the supply of US manufactured ketamine at a time when multiple suppliers of ketamine are advising the medical community that they are on backorder.
In addition to the pending ANDA application for Preservative Free Ketamine, NRXP is preparing a New Drug Application under Fast Track Designation to expand the use of intravenous ketamine to treat patients with severe depression, who may have suicidal ideation.
Strategic Partnership with Emobot to Integrate AI-Powered “Depression Thermometer” Across Interventional Psychiatry Network
On March 30th NRXP announced a strategic partnership with Emobt Health to deploy its revolutionary AI-driven emotional monitoring platform across the rapidly expanding NRXP network of interventional psychiatry clinics. This roll-out represents the first large-scale clinical integration of Emobot’s "Depression Thermometer," a passive monitoring tool designed to detect early treatment response and prevent relapse in patients with Treatment-Resistant Depression (TRD).
Emobot’s technology replaces cumbersome psychometric questionnaires with a background application on the user’s smartphone to passively and automatically measure levels of depression and potentially suicidality and PTSD. In recent years, there has been widespread recognition that neuroplastic therapies, including potentially IV Ketamine, SPRAVATO® (esketamine), and Transcranial Magnetic Stimulation (TMS) have the potential to replace an older and less effective generation of oral antidepressants.
Full-Year 2025 Results and Highlights Key Regulatory and Commercial Progress
On March 24th NRXP announced the filing of its Form 10-K for the year ended December 31, 2025. Highlights included the following key points:
A year-over-year reduction in operating expenses with $7.8 Million cash on hand at year end. With ongoing revenue from operations and current ATM trends, NRXP anticipates adequate resources to support operations through 2026, with anticipation of continued revenue growth. As of December 31, 2025, all previously issued convertible debt had been converted into common stock.
Anticipated FDA approval of at least one NRXP new drug – Preservative Free Ketamine under an ANDA—during Q3 2026. A favorable preliminary bioequivalence determination was received from the FDA.
A Type C in-person meeting with the FDA Division of Psychiatry Products together with leadership of the FDA Center for Drug Evaluation and Research (CDER) to identify a path to approval of NRXP NRX-100 (preservative-free ketamine) under a New Drug Application with an indication for treatment of Severe Depression in Patients who may have suicidality. In meeting minutes, FDA expressed willingness to review existing randomized clinical trial data and Real World Evidence as a potential basis for approval without a requirement for conduct of additional clinical trials.
Appointment of Prof. Joshua Brown, MD, PhD, of Harvard/Mclean as NRXP Chief Medical Innovation Officer. Dr. Brown is Principal Investigator on NIH- and DARPA-funded projects that highlight the future of neuroplastic care, including use of D-cycloserine and TMS for treating depression, PTSD, and suicidality.
Filing of an Investigational New Drug application with the FDA to study the use of the NRXP Breakthrough-designated candidate, NRX-101, for the enhancement of Transcranial Magnetic Stimulation (TMS) supported by non-dilutive governmental funding sources.
Development of a patentable, sustained-release presentation of D-cycloserine to provide an extended release profile suitable for enhancement of TMS efficacy. Prior NRXP clinical trials have shown a doubling of clinical response in patients with depression and an 8-fold increase in remission from depression versus standard TMS therapy.
Implementation of a business plan for NRXP subsidiary HOPE Therapeutics, Inc. with the aim of developing interventional psychiatry centers focused on neuroplastic therapy – including ketamine, D-cycloserine, Transcranial Magnetic Stimulation, and Hyperbaric Oxygen Therapy – for patients with severe and suicidal depression and PTSD, with future applications for Traumatic Brain Injury, Autism, and Alzheimer’s.
First NRXP revenue from acquired and partnered interventional psychiatry clinics at five sites of care that provide neuroplastic therapy to treat severe and suicidal depression and PTSD, with funding from the Department of Veterans Affairs, the Department of War, private insurers, and self-pay. Company expects to substantially increase this revenue-generating footprint in 2026.
Announcement of a nationwide NRXP partnership with neurocare AG, a leading manufacturer of TMS technology to develop their growing combined clinic base, currently at 20 sites of care in the US together with an installed base of more than 400 TMS machines into a nationwide accountable care network providing psychiatrist-led neuroplastic therapy.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Email: mduffy@nrxpharma.com
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Media Contact
Company Name: NRx Pharmaceuticals, Inc.
Contact Person: Matthew Duffy, Chief Business Officer
Email: Send Email
Phone: 484 254 6134
Address:1201 Orange Street Suite 600
City: Miami
State: Florida
Country: United States
Website: https://www.nrxpharma.com/








